Lasers in the past
2020 was a breakthrough in alternative treatments
The FDA Society has released sensational data on the
PlasmaHealthRegeneration method.
Comparative characteristics and uniqueness of low-temperature plasma (LTP) technology in aesthetic medicine: always one step ahead!
Plasma skin regeneration (PSR) is a new method of resurfacing that uses low-temperature plasma energy to create a thermal effect on the skin. PSR differs from lasers, light sources and ablative lasers in that it does not depend on chromophores and does not vaporize tissue, but leaves a layer of undamaged desiccated epidermis that acts as a natural biological bandage and promotes wound healing and rapid healing.
For a clearer understanding of the difference in the effects of the CO2 laser and the NTP device on the skin, we need to introduce two concepts: the zone of thermal damage - SST (directly sublimated areas) and the zone of thermal modification - SST (areas affected by partially dispersed, less dense energy).
In a study conducted on a live model, the results showed that treatment of pig skin with CO2 laser in different modes (light, medium, intensive) revealed a relatively larger zone of SQM, which contributed to a more prolonged healing than treatment with the NTP apparatus. Thus, due to the use of NTP apparatus for correction of skin imperfections, the clinician receives incomparably fast and high-quality wound healing and more intensive regeneration.
In addition, histological studies conducted in patients with plasma resurfacing confirmed continued collagen production, reduced elastosis, and progressive skin rejuvenation after 1 year of treatment. PSR (plasma skin resurfacing) has been listed by the US Food and Drug Administration 510(k) for the correction of facial and body wrinkles, superficial skin lesions, actinic keratoses, viral papillomas and seborrheic keratoses.
What are the features of plasma rejuvenation compared to laser rejuvenation, the alternative to which it was originally created?
NTP is also beneficial in the treatment of other conditions including dyschromia, photoaging, skin laxity, and post-acne scars. The safety profile of PSR is excellent, and there have been no reports of demarcation lines in the perioral, periorbital, or jaw regions, which can sometimes be seen after CO2 resurfacing. The Low-Temperature Plasmai method has been used for clinical studies and has been shown to be effective in correcting skin imperfections not only on the face, but also on the body, including the arms, neck, and chest. Numerous treatment protocols with adjustable energy settings allow for individualized treatments and provide the operator with precise control over the extent of injury and the length of the subsequent recovery period.
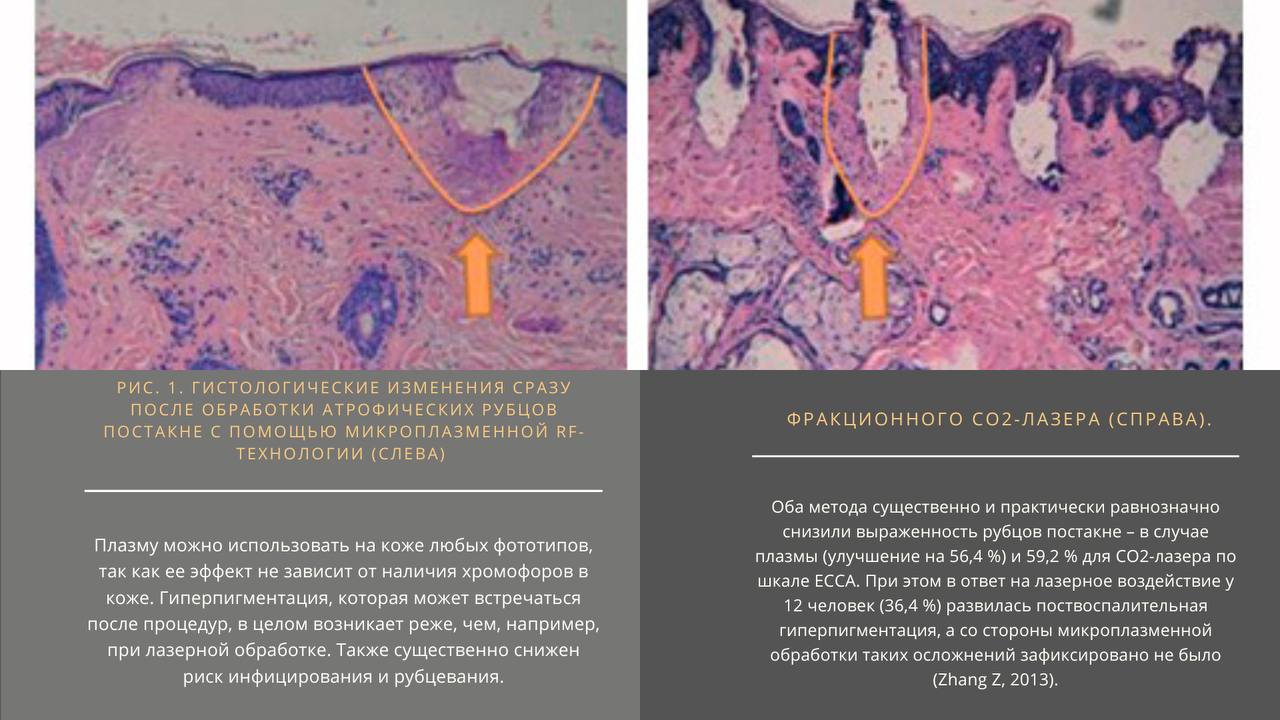
Histological changes immediately after treatment of atrophic postacne scars with microplasma technology (left) and fractionated CO2 laser (right).
The article was prepared by Oksana Astapieva, chief scientific officer of PlasmaHealth EDUCATION.
List of references:
A Histopathologic Evaluation of the Plasma Health
Regeneration System (PSR) Versus a Standard Carbon Dioxide Resurfacing Laser in an Animal Model
R. Fitzpatrick, MD,1* E. Bernstein, MD,2 S. Iyer, MD,3 D. Brown, PhD,2 P. Andrews,4 and K. Penny, MSc4, 1La Jolla Cosmetic Surgery Centre, San Diego, California
2 DakDak, LLC, Elkins Park, Pennsylvania
3 New York Dermatology Group, New York, New York
4 Rhytec Ltd, UK
Lasers in Surgery and Medicine 40:93–99 (2008)
Advanced in plasma health regeneration.
K Wade Foster, MD, PhD, Ronald L Moy, MD, & Edgar F Fincher, MD, PhD.
2008 Wiley Periodicals, Inc. •
Journal of Cosmetic Dermatology , 169–179
2008 Wiley Periodicals, Inc. •
Journal of Cosmetic Dermatology 169–179
Journal of Cosmetic Dermatology, 2008, Is. 7, p. p. 169-179.
Plasma health resurfacing for regeneration of neck, chest, and hands: investigation of a novel device.
Alster TS, Konda S.Dermatol Surg. 2007 Nov;33(11):1315-21. doi: 10.1111/j.1524-4725.2007.33282.x.
Evaluation of plasma health regeneration technology in low-energy full-facial rejuvenation
Melissa A Bogle 1, Kenneth A Arndt, Jeffrey S Dover
PMID: 17309997 DOI: 10.1001/archderm.143.2.168